 
As a result of the formation of two bonds, we will have a double bond structure. Step 4: The octet fulfilment idea is complete. Therefore, we can take the electrons from the bottom and position them in the middle between the two C atoms. However, the second carbon atom in the centre lacks two electrons. Here, we can observe that the octet of one carbon atom is complete (the Octet rule has been discussed before). Step 3: Now that the atoms have been represented by their symbols, let’s represent the valence electrons with dots. Hydrogen atoms will occupy the outermost locations. 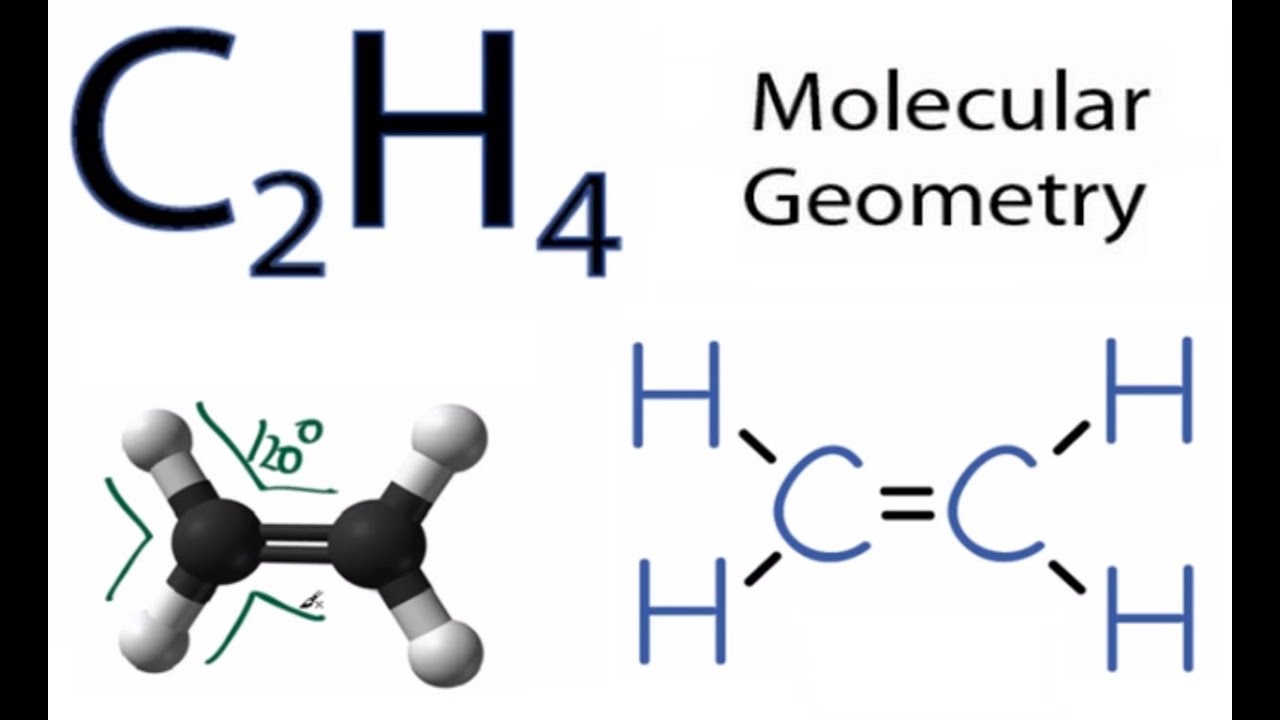
Step 2: Having determined the total valence number, we can now determine which atom is the least electronegative.įor hydrocarbons, the carbon will always be placed in the centre. 
The number of valence electrons in a single C2H4 molecule. In the case of carbon, each valence electron has four electrons. Hydrogen is the first element in the periodic table hence, it possesses one valence electron. Step 1: Count the number of atoms in an ethylene molecule. Obtaining the Lewis Structure of any molecule is simple if specific protocols are followed. This is referred to as the octet rule or octet completion. In order to achieve the same valency of eight, the atoms of the major groups tend to acquire extra electrons. Observing the final group reveals that all elements are inert gases with eight electrons in their valence shells (except He which has two). The number of an atom’s valence electrons is comparable to its valency, which in turn defines the atom’s combining capability. The outermost shell is known as the valence shell, and its electrons are known as valence electrons. The nucleus of an atom is around by negatively charged electrons, which are present at various levels or shells. The electron dot structure, also known as the Lewis Structure, is a diagrammatic representation of a molecule that includes the constituent atoms and valence shell electrons.īefore we dive in, we’d like to expose you to (or, if you’re already familiar, refresh you on) some incredibly key ideas that will make your knowledge of ethylene bonding much easier! Valence electrons Let’s proceed and discuss this systematically. The nature of this carbon-hydrogen bond is covalent. To gain a thorough understanding of ethylene, it is sufficient to first comprehend its bonding characteristics. We obtain molecular compounds based on the nature of atoms and their propensity to attract or repulsion another atom of the same or a different type.Īnd this process of two or more atoms approaching one another and deciding to remain together is known as chemical bonding. Now, atoms do not typically exist in a solitary state everything we observe around us is primarily composed of atoms that have merged to form molecules. How is this entire cosmos composed of several atoms? Have you ever pondered the universe’s singularity and immense variety? These hydrocarbons create double and triple bonds and are referred to as alkenes and alkynes, respectively. %20lewis%20structure.jpg)
Saturated hydrocarbons have the structure H-(CH2)n and are also known as acyclic straight-chain alkanes. Straight-chain hydrocarbons are the most basic and easiest to interpret group. There are various types of hydrocarbons in organic chemistry, including straight-chain, cyclic, and even branched hydrocarbons. Chemical Interactions in HydrocarbonsĬarbon has a covalent character when it comes to forming bonds with hydrogen, which results in the development of various types of hydrocarbons.įrom the simplest, such as methane and benzene, to the more complex, such as natural rubber, we encounter several HCs in our daily lives. Therefore, it is essential that we gain a thorough understanding of C2H4 to better comprehend the nature of straight-chain hydrocarbons. We have encountered this chemical whenever we have read about organic chemistry, haven’t we? This article will discuss ethylene, one of the most prevalent and commonly utilised hydrocarbons (C2H4).ĭo you realise this substance is even lighter than air?Ĭ2H4 is a simple straight-chain hydrocarbon with a sweet odour and a colourless appearance. Occasionally, it also produces compounds with additional elements, such as sulphur, nitrogen, etc.Įven though these are among of the simplest organic compounds we might encounter, their physical and chemical properties vary considerably. Additionally, they can be found in synthetic polymers and other man-made plastics.Īs the name suggests, they are organic in origin and composed of only carbon and hydrogen. The majority of hydrocarbons are found naturally in these fossil fuels, be they petroleum, crude oil, or natural gas. Hydrocarbons are an indispensable and inseparable component of chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
